Essential processes in mammalian cells are controlled by proteins called transcription factors. For example, the transcription factor HIF-1 is triggered by a low-oxygen situation to cause the cell to adapt to decreased oxygen.
Transcription factors operate in healthy cells, but cancer cells can co-opt transcription factors such as HIF-1 into promoting tumor growth.
New research from the lab of Hening Lin, professor of chemistry and chemical biology in the College of Arts and Sciences, finds that a protein called TiPARP acts as a terminator for several cancer-causing transcription factors, including HIF-1, which is implicated in many cancers, including breast cancer. The research demonstrates that TiPARP, therefore, is a tumor suppressor.
The paper “TiPARP Forms Nuclear Condensates to Degrade HIF-1α and Suppress Tumorigenesis,” published in PNAS, establishes TiPARP as a turning-off mechanism for several important transcription factors – including HIF-1, C0-Myc and estrogen receptor – and shows how TiPARP itself is degraded during this process. The study also shows the mechanism through which TiPARP terminates these factors, another new discovery.
Co-authors are graduate student Lu Zhang; former postdoctoral fellow Ji Cao; and Longying Dong, former director of the Immunopathology Research and Development Laboratory in the College of Veterinary Medicine’s Department of Biomedical Sciences.
HIF-1 is important for cancer because a lot of tumors thrive in low-oxygen conditions, said Lin, a Howard Hughes Medical Institute Investigator and corresponding author of the paper.
“For these tumors to survive, they have to rely on HIF-1,” Lin said. “TiPARP is a terminator of HIF-1. Therefore, if you can activate TiPARP, then you can suppress [tumor growth].”
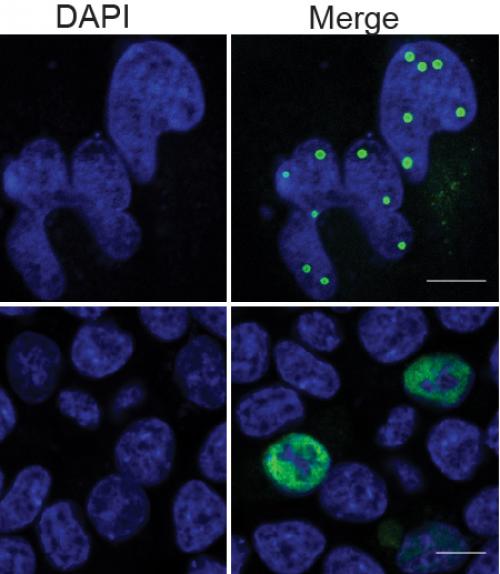
Lin and co-authors are also excited by the discovery of the mechanism through which TiPARP brings about the termination of HIF-1 and other transcription factors. This mechanism, called “liquid-liquid phase separation” or “phase condensation,” is a topic of great interest in biology.
Imagine drops of vinegar in oil: The vinegar forms distinct droplets suspended in the more viscous oil.
Similarly, when TiPARP is activated in a cell nucleus, it forms the so-called “phase separation” that recruits HIF-1-alpha and HUWE1 (an ubiquitin protein ligase) in the cell nucleus. This starts a process through which HIF-1-alpha and TiPARP both deactivate and degrade.
Through phase separation, TiPARP terminates not just HIF-1 but several different transcription factors implicated in different types of cancer, Lin said.
In fact, TiPARP may already be at work in Tamoxifen, a widely used breast cancer drug. Lin thinks Tamoxifen, which successfully treats estrogen receptor-positive breast cancers, works because TiPARP is actively terminating estrogen receptor, HIF-1 and c-Myc in tumors.
“When Tamoxifen and similar compounds were developed as estrogen receptor agonists or antagonists, we did not even know what compounds would be better,” he said. “Now we think the idea will be, what compound can activate TiPARP better?”
This research received support from the Howard Hughes Medical Institute, Cornell’s College of Arts and Sciences, and the National Institutes of Health.
Read the story in the Cornell Chronicle.